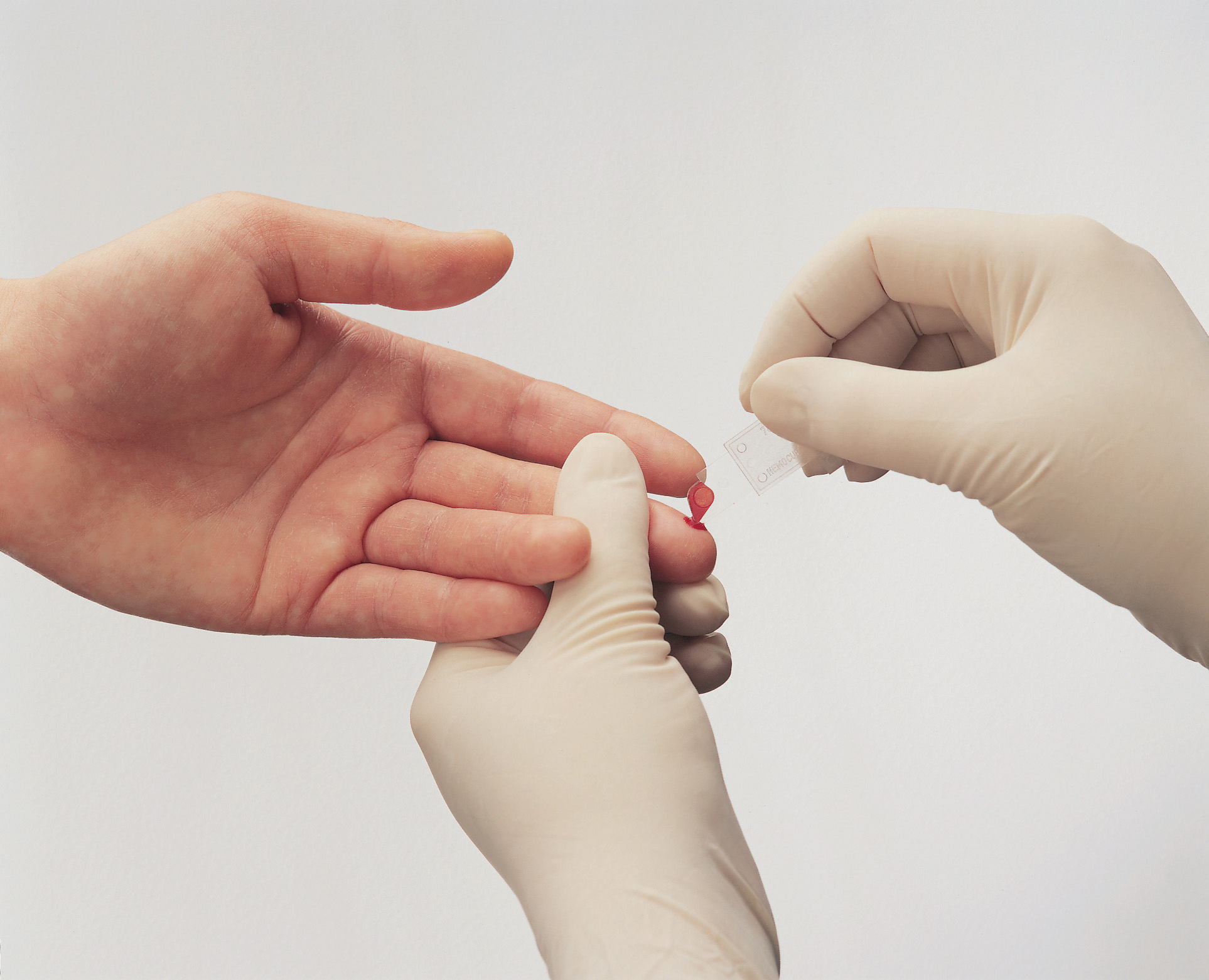
We are HemoCue,
we advance care.
Who we are
For more than four decades we’ve been advancing what’s possible at the point of care.
Our caring commitment to our people, partners and patients remains as strong as it was on the first day of our journey.

What we do
We provide foundational diagnostic solutions that enable high-quality care across clinical settings and geographies.
Standing on time tested expertise we work in close collaboration with stakeholders across the healthcare industry to transform diagnostics data into insights that enhance global health.
HemoCue is proud to be part of Danaher, a global life sciences and diagnostics innovator committed to accelerating the power of science and technology to improve human health.
As part of Danaher, HemoCue is connected to a uniquely broad ecosystem of expertise, technologies, and capabilities. Together, we partner closely with customers to solve their toughest challenges with greater speed and certainty.
Powered by the rigor of the Danaher Business System and a culture of continuous improvement, we make time a competitive advantage for our customers and help unlock the transformative potential of science and technology to improve billions of lives every day.
Purpose
Advancing care,
test by test,
everywhere
Healthcare today faces tough challenges, affecting both providers and patients. But through the waves of rising burdens, evolving technology and system reforms we dare to make it simple and say, that the essence of Healthcare is Care.
Care is the bedrock upon which we stand. It’s manifested in our devices, our people and how we interact with the world. While our world revolves around a drop of blood, we strive to be a foundational force that advances care one test at a time.
HemoCue in numbers
Tests performed per year
across disease areas
Countries served
across the globe
Global employees
committed to care
Years of innovation
fuelled by engineering excellence
The HemoCue story

“It must be possible”
This conviction has been the driving force behind HemoCue since day one. The day, over 40 years ago, when two men said “It must be possible”. To do this easier. To do this better. To do this right.
What the inventors, Jan Lilja and Sven-Erik Nilsson, saw as they visited labs and doctors’ offices triggered the spark that drives human invention: the ability to see possibilities where others see problems. They saw the frustration within healthcare of having to wait to treat patients or adjust test values that were always 30-40% off. They saw the inaccuracy and dangers of limited reagent durability, shifting instrument calibration, and pipetting. The impracticality of the time it took to send samples to the lab – so troublesome that healthcare workers preferred to send the patient instead.
They recognized that addressing these issues would simplify tasks for physicians, nurses, and labs, and could potentially save lives. There was no doubt. It had to be done. And it had to be done right. It took time and involved much effort. After all, they were working with an exact science inside a microworld. One that had to be repeatable, yielding the same value – the right value – time after time. It was difficult and often frustrating. Seemingly at every turn, they were told, “It’s impossible”.
Yet, they never gave up. Never compromised. And achieved something remarkable — a point-of-care blood test with tolerances never before thought possible, let alone repeatable with accuracy in tens of millions of cuvettes per year.
And that was only the beginning. Their problem-solving focus and drive have been instilled in the hearts and minds of everyone in the HemoCue family since then. With a positive, open-minded atmosphere, HemoCue continues to foster a belief in possibilities and a dedication to getting it right. If there’s an issue or challenge, it’s a concern for the entire company, not just one person. Everyone is committed to resolving it because every detail matters to our customers. It’s that “can do” team spirit that has led to a continuous stream of game-changing innovations over the past 40 years.
Today, the impossible has become the standard. 4 HemoCue tests are performed each second worldwide. That means by the time you finish reading this, another 480 people will have the answers they need at the point of care.
But we won’t rest. We always have another challenge in our sights. And we say, as they did, “It must be possible”.